.

Worlds Within Worlds:
The Story of Nuclear Energy
Volume 2
Mass and Energy · The Neutron · The Structure of the Nucleus
by Isaac Asimov
U. S. Energy Research and Development Administration
Office of Public Affairs
Washington, D.C. 20545
Library of Congress Catalog Card Number: 75-189477
1972
Nothing in the history of mankind has opened our eyes to the possibilities of science as has the development of atomic power. In the last 200 years, people have seen the coming of the steam engine, the steamboat, the railroad locomotive, the automobile, the airplane, radio, motion pictures, television, the machine age in general. Yet none of it seemed quite so fantastic, quite so unbelievable, as what man has done since 1939 with the atom ... there seem to be almost no limits to what may lie ahead: inexhaustible energy, new worlds, ever-widening knowledge of the physical universe. Isaac Asimov

The U. S. Energy Research and Development Administration publishes a series of booklets for the general public.
Please write to the following address for a title list or for information on a specific subject:
USERDA—Technical Information Center
P. O. Box 62
Oak Ridge, Tennessee 37830

ISAAC ASIMOV received his academic degrees from Columbia University and is Associate Professor of Biochemistry at the Boston University School of Medicine. He is a prolific author who has written over 150 books in the past 20 years, including about 20 science fiction works, and books for children. His many excellent science books for the public cover subjects in mathematics, physics, astronomy, chemistry, and biology, such as The Genetic Code, Inside the Atom, Building Blocks of the Universe, Understanding Physics, The New Intelligent Man’s Guide to Science, and Asimov’s Biographical Encyclopedia of Science and Technology.
In 1965 Dr. Asimov received the James T. Grady Award of the American Chemical Society for his major contribution in reporting science progress to the public.

- VOLUME 1
- Introduction 5
- Atomic Weights 6
- Electricity 11
- Units of Electricity 11
- Cathode Rays 13
- Radioactivity 17
- The Structure of the Atom 25
- Atomic Numbers 30
- Isotopes 35
- Energy 47
- The Law of Conservation of Energy 47
- Chemical Energy 50
- Electrons and Energy 54
- The Energy of the Sun 55
- The Energy of Radioactivity 57
- VOLUME 2
- Mass and Energy 69
- The Structure of the Nucleus 75
- The Proton 75
- The Proton-Electron Theory 76
- Protons in Nuclei 80
- Nuclear Bombardment 82
- Particle Accelerators 86
- The Neutron 92
- Nuclear Spin 92
- Discovery of the Neutron 95
- The Proton-Neutron Theory 98
- The Nuclear Interaction 101
- Neutron Bombardment 107
- VOLUME 3
- Nuclear Fission 117
- New Elements 117
- The Discovery of Fission 122
- The Nuclear Chain Reaction 127
- The Nuclear Bomb 131
- Nuclear Reactors 141
- Nuclear Fusion 147
- The Energy of the Sun 147
- Thermonuclear Bombs 149
- Controlled Fusion 151
- Beyond Fusion 159
- Antimatter 159
- The Unknown 164
- Reading List 166

A field-ion microscope view of atoms in a crystal. Each tiny white dot is a single atom, and each ring system is a crystal facet or plane. The picture is magnified 1,500,000 times.
MASS AND ENERGY
In 1900 it began to dawn on physicists that there was a vast store of energy within the atom; a store no one earlier had imagined existed. The sheer size of the energy store in the atom—millions of times that known to exist in the form of chemical energy—seemed unbelievable at first. Yet that size quickly came to make sense as a result of a line of research that seemed, at the beginning, to have nothing to do with energy.
Suppose a ball were thrown forward at a velocity of 20 kilometers per hour by a man on top of a flatcar that is moving forward at 20 kilometers an hour. To someone watching from the roadside the ball would appear to be travelling at 40 kilometers an hour. The velocity of the thrower is added to the velocity of the ball.
If the ball were thrown forward at 20 kilometers an hour by a man on top of a flatcar that is moving backward at 20 kilometers an hour, then the ball (to someone watching from the roadside) would seem to be not moving at all after it left the hand of the thrower. It would just drop to the ground.
There seemed no reason in the 19th century to suppose that light didn’t behave in the same fashion. It was known to travel at the enormous speed of just a trifle under 300,000 kilometers per second, while earth moved in its orbit about the sun at a speed of about 30 kilometers per second. Surely if a beam of light beginning at some earth-bound source shone in the direction of earth’s travel, it ought to move at a speed of 300,030 kilometers per second. If it shone in the opposite direction, against earth’s motion, it ought to move at a speed of 299,970 kilometers per second.
Could such a small difference in an enormous speed be detected?

Albert A. Michelson
The German-American physicist Albert Abraham Michelson (1852-1931) had invented a delicate instrument, the interferometer, that could compare the velocities of different beams of light with great precision. In 1887 he and a co-worker, the American chemist Edward Williams Morley (1838-1923), tried to measure the comparative speeds of light, using beams headed in different directions. Some of this work was performed at the U. S. Naval Academy and some at the Case Institute.
The results of the Michelson-Morley experiment were unexpected. It showed no difference in the measured speed of light. No matter what the direction of the beam—whether it went in the direction of the earth’s movement, or against it, or at any angle to it—the speed of light always appeared to be exactly the same.
To explain this, the German-Swiss-American scientist Albert Einstein (1879-1955) advanced his “special theory of relativity” in 1905. According to Einstein’s view, speeds could not merely be added. A ball thrown forward at 20 kilometers an hour by a man moving at 20 kilometers an hour in the same direction would not seem to be going 40 kilometers an hour to an observer at the roadside. It would seem to be going very slightly less than 40 kilometers an hour; so slightly less that the difference couldn’t be measured.
However, as speeds grew higher and higher, the discrepancy in the addition grew greater and greater (according to a formula Einstein derived) until, at velocities of tens of thousands of kilometers per hour, that discrepancy could be easily measured. At the speed of light, which Einstein showed was a limiting velocity that an observer would never reach, the discrepancy became so great that the speed of the light source, however great, added or subtracted zero to or from the speed of light.
Accompanying this were all sorts of other effects. It could be shown by Einstein’s reasoning that no object possessing mass could move faster than the speed of light. What’s more, as an object moved faster and faster, its length in the direction of motion (as measured by a stationary 72 observer) grew shorter and shorter, while its mass grew greater and greater. At 260,000 kilometers per second, its length in the direction of movement was only half what it was at rest, and its mass was twice what it was. As the speed of light was approached, its length would approach zero in the direction of motion, while its mass would approach the infinite.
Could this really be so? Ordinary objects never moved so fast as to make their lengths and masses show any measurable change. What about subatomic particles, however, which moved at tens of thousands of kilometers per second? The German physicist Alfred Heinrich Bucherer (1863-1927) reported in 1908 that speeding electrons did gain in mass just the amount predicted by Einstein’s theory. The increased mass with energy has been confirmed with great precision in recent years. Einstein’s special theory of relativity has met many experimental tests exactly ever since and it is generally accepted by physicists today.
Einstein’s theory gave rise to something else as well. Einstein deduced that mass was a form of energy. He worked out a relationship (the “mass-energy equivalence”) that is expressed as follows:
E = mc²
where E represents energy, m is mass, and c is the speed of light.
If mass is measured in grams and the speed of light is measured in centimeters per second, then the equation will yield the energy in a unit called “ergs”. It turns out that 1 gram of mass is equal to 900,000,000,000,000,000,000 (900 billion billion) ergs of energy. The erg is a very small unit of energy, but 900 billion billion of them mount up.
The energy equivalent of 1 gram of mass (and remember that a gram, in ordinary units, is only ¹/₂₈ of an ounce) would keep a 100-watt light bulb burning for 35,000 years.
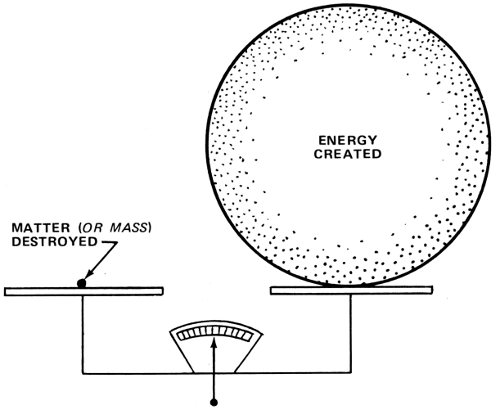
It is this vast difference between the tiny quantity of mass and the huge amount of energy to which it is equivalent that obscured the relationship over the years. When a chemical reaction liberates energy, the mass of the materials undergoing the reaction decreases slightly—but very slightly.
Suppose, for instance, a gallon of gasoline is burned. The gallon of gasoline has a mass of 2800 grams and combines with about 10,000 grams of oxygen to form carbon dioxide and water, yielding 1.35 million billion ergs. That’s a lot of energy and it will drive an automobile for some 25 to 30 kilometers. But by Einstein’s equation all that energy is equivalent to only a little over a millionth of a gram. You start with 12,800 grams of reacting materials and you end with 12,800 grams minus a millionth of a gram or so that was given off as energy.
No instrument known to the chemists of the 19th century could have detected so tiny a loss of mass in such a 74 large total. No wonder, then, that from Lavoisier on, scientists thought that the law of conservation of mass held exactly.
Radioactive changes gave off much more energy per atom than chemical changes did, and the percentage loss in mass was correspondingly greater. The loss of mass in radioactive changes was found to match the production of energy in just the way Einstein predicted.
It was no longer quite accurate to talk about the conservation of mass after 1905 (even though mass was just about conserved in ordinary chemical reactions so that the law could continue to be used by chemists without trouble). Instead, it is more proper to speak of the conservation of energy, and to remember that mass was one form of energy and a very concentrated form.
The mass-energy equivalence fully explained why the atom should contain so great a store of energy. Indeed, the surprise was that radioactive changes gave off as little energy as they did. When a uranium atom broke down through a series of steps to a lead atom, it produced a million times as much energy as that same atom would release if it were involved in even the most violent of chemical changes. Nevertheless, that enormous energy change in the radioactive breakdown represented only about one-half of 1% of the total energy to which the mass of the uranium atom was equivalent.
Once Rutherford worked out the nuclear theory of the atom, it became clear from the mass-energy equivalence that the source of the energy of radioactivity was likely to be in the atomic nucleus where almost all the mass of the atom was to be found.
The attention of physicists therefore turned to the nucleus.
THE STRUCTURE OF THE NUCLEUS
The Proton
As early as 1886 Eugen Goldstein, who was working with cathode rays, also studied rays that moved in the opposite direction. Since the cathode rays (electrons) were negatively charged, rays moving in the opposite direction would have to be positively charged. In 1907 J. J. Thomson called them “positive rays”.
Once Rutherford worked out the nuclear structure of the atom, it seemed clear that the positive rays were atomic nuclei from which a number of electrons had been knocked away. These nuclei came in different sizes.
Were the nuclei single particles—a different one for every isotope of every element? Or were they all built up out of numbers of still smaller particles of a very limited number of varieties? Might it be that the nuclei owed their positive electrical charge to the fact that they contained particles just like the electron, but ones that carried a positive charge rather than a negative one?
All attempts to discover this “positive electron” in the nuclei failed, however. The smallest nucleus found was that produced by knocking the single electron off a hydrogen atom in one way or another. This hydrogen nucleus had a single positive charge, one that was exactly equal in size to the negative charge on the electron. The hydrogen nucleus, however, was much more massive than an electron. The hydrogen nucleus with its single positive charge was approximately 1837 times as massive as the electron with its single negative charge.
Was it possible to knock the positive charge loose from the mass of the hydrogen nucleus? Nothing physicists did could manage to do that. In 1914 Rutherford decided the attempt should be given up. He suggested that the hydrogen nucleus, for all its high mass, should be considered the unit of 76 positive electrical charge, just as the electron was the unit of negative electrical charge. He called the hydrogen nucleus a “proton” from the Greek word for “first” because it was the nucleus of the first element.
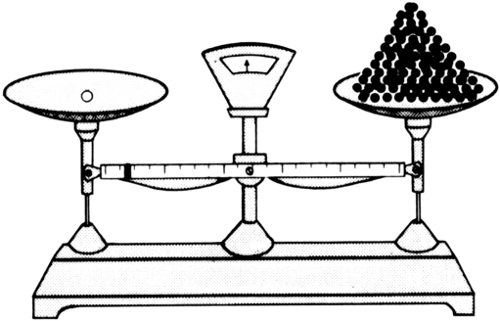
One proton balances 1837 electrons.
Why the proton should be so much more massive than the electron is still one of the unanswered mysteries of physics.
The Proton-Electron Theory
What about the nuclei of elements other than hydrogen?
All the other elements had nuclei more massive than that of hydrogen and the natural first guess was that these were made up of some appropriate number of protons closely packed together. The helium nucleus, which had a mass four times as great as that of hydrogen, might be made up of 4 protons; the oxygen nucleus with a mass number of 16 might be made up of 16 protons and so on.
This guess, however, ran into immediate difficulties. A helium nucleus might have a mass number of 4 but it had an electric charge of +2. If it were made up of 4 protons, it ought to have an electric charge of +4. In the same way, an 77 oxygen nucleus made up of 16 protons ought to have a charge of +16, but in actual fact it had one of +8.
Could it be that something was cancelling part of the positive electric charge? The only thing that could do so would be a negative electric charge[1] and these were to be found only on electrons as far as anyone knew in 1914. It seemed reasonable, then, to suppose that a nucleus would contain about half as many electrons in addition to the protons. The electrons were so light, they wouldn’t affect the mass much, and they would succeed in cancelling some of the positive charge.
Thus, according to this early theory, now known to be incorrect, the helium nucleus contained not only 4 protons, but 2 electrons in addition. The helium nucleus would then have a mass number of 4 and an electric charge (atomic number) of 4 - 2, or 2. This was in accordance with observation.
This “proton-electron theory” of nuclear structure accounted for isotopes very nicely. While oxygen-16 had a nucleus made up of 16 protons and 8 electrons, oxygen-17 had one of 17 protons and 9 electrons, and oxygen-18 had one of 18 protons and 10 electrons. The mass numbers were 16, 17, and 18, respectively, but the atomic number was 16 - 8, 17 - 9, and 18 - 10, or 8 in each case.
Again, uranium-238 has a nucleus built up, according to this theory, of 238 protons and 146 electrons, while uranium-235 has one built up of 235 protons and 143 electrons. In these cases the atomic number is, respectively, 238 - 146 and 235 - 143, or 92 in each case. The nucleus of the 2 isotopes is, however, of different structure and it is not surprising therefore that the radioactive properties of the 78 two—properties that involve the nucleus—should be different and that the half-life of uranium-238 should be six times as long as that of uranium-235.
The presence of electrons in the nucleus not only explained the existence of isotopes, but seemed justified by two further considerations.
First, it is well known that similar charges repel each other and that the repulsion is stronger the closer together the similar charges are forced. Dozens of positively charged particles squeezed into the tiny volume of an atomic nucleus couldn’t possibly remain together for more than a tiny fraction of a second. Electrical repulsion would send them flying apart at once.
On the other hand, opposite charges attract, and a proton and an electron would attract each other as strongly as 2 protons (or 2 electrons) would repel each other. It was thought possible that the presence of electrons in a collection of protons might somehow limit the repulsive force and stabilize the nucleus.
Second, there are radioactive decays in which beta particles are sent flying out of the atom. From the energy involved they could come only out of the nucleus. Since beta particles are electrons and since they come from the nucleus, it seemed to follow that there must be electrons within the nucleus to begin with.
The proton-electron theory of nuclear structure also seemed to account neatly for many of the facts of radioactivity.
Why radioactivity at all, for instance? The more complex a nucleus is, the more protons must be squeezed together and the harder, it would seem, it must be to keep them together. More and more electrons seemed to be required. Finally, when the total number of protons was 84 or more, no amount of electrons seemed sufficient to stabilize the nucleus.
The manner of breakup fits the theory, too. Suppose a nucleus gives off an alpha particle. The alpha particle is a helium nucleus made up, by this theory, of 4 protons and 2 electrons. If a nucleus loses an alpha particle, its mass number should decline by 4 and its atomic number by 4 - 2, or 2. And, indeed, when uranium-238 (atomic number 92) gives off an alpha particle, it becomes thorium-234 (atomic number 90).
Suppose a beta particle is emitted. A beta particle is an electron and if a nucleus loses an electron, its mass number is almost unchanged. (An electron is so light that in comparison with the nucleus, we can ignore its mass.) On the other hand, a unit negative charge is gone. One of the protons in the nucleus, which had previously been masked by an electron, is now unmasked. Its positive charge is added to the rest and the atomic number goes up by one. Thus, thorium-234 (atomic number 90) gives up a beta particle and becomes protactinium-234 (atomic number 91).
If a gamma ray is given off, that gamma ray has no charge and the equivalent of very little mass. That means that neither the mass number nor the atomic number of the nucleus is changed, although its energy content is altered.
Even more elaborate changes can be taken into account. In the long run, uranium-238, having gone through many changes, becomes lead-206. Those changes include the emission of 8 alpha particles and 6 beta particles. The 8 alpha particles involve a loss of 8 × 4, or 32 in mass number, while the 6 beta particles contribute nothing in this respect. And, indeed, the mass number of uranium-238 declines by 32 in reaching lead-206. On the other hand the 8 alpha particles involve a decrease in atomic number of 8 × 2, or 16, while the 6 beta particles involve an increase in atomic number of 6 × 1, or 6. The total change is a decrease of 16 - 6, or 10. And indeed, uranium (atomic number 92) changes to lead (atomic number 82).
It is useful to go into such detail concerning the proton-electron theory of nuclear structure and to describe how attractive it seemed. The theory appeared solid and unshakable and, indeed, physicists used it with considerable satisfaction for 15 years.
—And yet, as we shall see, it was wrong; and that should point a moral. Even the best seeming of theories may be wrong in some details and require an overhaul.
Protons in Nuclei
Let us, nevertheless, go on to describe some of the progress made in the 1920s in terms of the proton-electron theory that was then accepted.
Since a nucleus is made up of a whole number of protons, its mass ought to be a whole number if the mass of a single proton is considered 1. (The presence of electrons would add some mass but in order to simplify matters, let us ignore that.)
When isotopes were first discovered this indeed seemed to be so. However, Aston and his mass spectrometer kept measuring the mass of different nuclei more and more closely during the 1920s and found that they differed very slightly from whole numbers. Yet a fixed number of protons turned out to have different masses if they were first considered separately and then as part of a nucleus.
Using modern standards, the mass of a proton is 1.007825. Twelve separate protons would have a total mass of twelve times that, or 12.0939. On the other hand, if the 12 protons are packed together into a carbon-12 nucleus, the mass is 12 so that the mass of the individual protons is 1.000000 apiece. What happens to this difference of 0.007825 between the proton in isolation and the proton as part of a carbon-12 nucleus?
According to Einstein’s special theory of relativity, the missing mass would have to appear in the form of energy. If 12 hydrogen nuclei (protons) plus 6 electrons are packed 81 together to form a carbon nucleus, a considerable quantity of energy would have to be given off.
In general, Aston found that as one went on to more and more complicated nuclei, a larger fraction of the mass would have to appear as energy (though not in a perfectly regular way) until it reached a maximum in the neighborhood of iron.
Iron-56, the most common of the iron isotopes, has a mass number of 55.9349. Each of its 56 protons, therefore, has a mass of 0.9988.
For nuclei more complicated than those of iron, the protons in the nucleus begin to grow more massive again. Uranium-238 nuclei, for instance, have a mass of 238.0506, so that each of the 238 protons they contain has a mass of 1.0002.
By 1927 Aston had made it clear that it is the middle elements in the neighborhood of iron that are most closely and economically packed. If a very massive nucleus is broken up into somewhat lighter nuclei, the proton packing would be tighter and some mass would be converted into energy. Similarly, if very light nuclei were joined together into somewhat more massive nuclei, some mass would be converted into energy.
This demonstration that energy was released in any shift away from either extreme of the list of atoms according to atomic number fits the case of radioactivity, where very massive nuclei break down to somewhat less massive ones.
Consider that uranium-238 gives up 8 alpha particles and 6 beta particles to become lead-206. The uranium-238 nucleus has a mass of 238.0506; each alpha particle has one of 4.0026 for a total of 32.0208; each beta particle has a mass of 0.00154 for a total of 0.00924; and the lead-206 nucleus has one of 205.9745.
This means that the uranium-238 nucleus (mass: 238.0506) changes into 8 alpha particles, 6 beta particles, and a lead-206 nucleus (total mass: 238.0045). The starting 82 mass is 0.0461 greater than the final mass and it is this missing mass that has been converted into energy and is responsible for the gamma rays and for the velocity with which alpha particles and beta particles are discharged.
Nuclear Bombardment
Once scientists realized that there was energy which became available when one kind of nucleus was changed into another, an important question arose as to whether such a change could be brought about and regulated by man and whether this might not be made the source of useful power of a kind and amount undreamed of earlier.
Chemical energy was easy to initiate and control, since that involved the shifts of electrons on the outskirts of the atoms. Raising the temperature of a system, for instance, caused atoms to move more quickly and smash against each other harder, and that in itself was sufficient to force electrons to shift and to initiate a chemical reaction that would not take place at lower temperatures.
To shift the protons within the nucleus (“nuclear reactions”) and make nuclear energy available was a harder problem by far. The particles involved were much more massive than electrons and correspondingly harder to move. What’s more, they were buried deep within the atom. No temperatures available to the physicists of the 1920s could force atoms to smash together hard enough to reach and shake the nucleus.
In fact, the only objects that were known to reach the nucleus were speeding subatomic particles. As early as 1906, for instance, Rutherford had used the speeding alpha particles given off by a radioactive substance to bombard matter and to show that sometimes these alpha particles were deflected by atomic nuclei. It was, in fact, by such an experiment that he first demonstrated the existence of such nuclei.
Rutherford had continued his experiments with bombardment. An alpha particle striking a nucleus would knock it free of the atom to which it belonged and send it shooting forward (like one billiard ball hitting another). The nucleus that shot ahead would strike a film of chemical that scintillated (sparkled) under the impact. In a rough way, one could tell the kind of nucleus that struck from the nature of the sparkling.
In 1919 Rutherford bombarded nitrogen gas with alpha particles and found that he obtained the kind of sparkling he associated with the bombardment of hydrogen gas. When he bombarded hydrogen, the alpha particles struck hydrogen nuclei (protons) and shot them forward. To get hydrogen-sparkling out of the bombardment of nitrogen, Rutherford felt, he must have knocked protons out of the nitrogen nuclei. Indeed, as was later found, he had converted nitrogen nuclei into oxygen nuclei.
This was the first time in history that the atomic nucleus was altered by deliberate human act.
Rutherford continued his experiments and by 1924 had shown that alpha particles could be used to knock protons out of the nuclei of almost all elements up to potassium (atomic number 19).
There were, however, limitations to the use of natural alpha particles as the bombarding agent.
First, the alpha particles used in bombardment were positively charged and so were the atomic nuclei. This meant that the alpha particles and the atomic nuclei repelled each other and much of the energy of the alpha particles was used in overcoming the repulsion. For more and more massive nuclei, the positive charge grew higher and the repulsion stronger until for elements beyond potassium, no collision could be forced, even with the most energetic naturally occurring alpha particles.
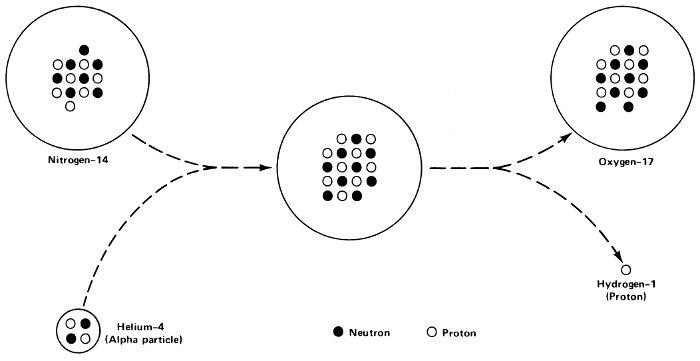
Man-made transmutation.
Second, the alpha particles that are sprayed toward the target cannot be aimed directly at the nuclei. An alpha particle strikes a nucleus only if, by chance, they come together. The nuclei that serve as their targets are so unimaginably small that most of the bombarding particles are sure to miss. In Rutherford’s first bombardment of nitrogen, it was calculated that only 1 alpha particle out of 300,000 managed to strike a nitrogen nucleus.
The result of these considerations is clear. There is energy to be gained out of nuclear reactions, but there is also energy that must be expended to cause these nuclear reactions. In the case of nuclear bombardment by subatomic particles (the only way, apparently, in which nuclear reactions can be brought about), the energy expended seems to be many times the energy to be extracted. This is because so many subatomic particles use up their energy in ionizing atoms, knocking electrons away, and never initiate nuclear reactions at all.
It was as though the only way you could light a candle would be to strike 300,000 matches, one after the other. If that were so, candles would be impractical.
In fact, the most dramatic result of alpha particle bombardment had nothing to do with energy production, but rather the reverse. New nuclei were produced that had more energy than the starting nuclei, so that energy was absorbed by the nuclear reaction rather than given off.
This came about first in 1934, when a French husband-and-wife team of physicists, Frédéric Joliot-Curie (1900-1958) and Irène Joliot-Curie (1897-1956) were bombarding aluminum-27 (atomic number 13) with alpha particles. The result was to combine part of the alpha particle with the aluminum-27 nucleus to form a new nucleus with an atomic number two units higher—15—and a mass number three units higher—30.
The element with atomic number 15 is phosphorus so that phosphorus-30 was formed. The only isotope of phosphorus 86 that occurs in nature, however, is phosphorus-31. Phosphorus-30 was the first man-made nucleus—the first to be manufactured by nuclear reactions in the laboratory.

Frédéric and Irène Joliot-Curie
The reason phosphorus-30 did not occur in nature was that its energy content was too high to allow it to be stable. Its energy content drained away through the emission of particles that allowed the nucleus to change over into a stable one, silicon-30 (atomic number 14). This was an example of “artificial radioactivity”.
Since 1934, over a thousand kinds of nuclei that do not occur in nature have been formed in the laboratory through various kinds of bombardment-induced nuclear reactions. Every single one of them proved to be radioactive.
Particle Accelerators
Was there nothing that could be done to make nuclear bombardment more efficient and increase the chance of obtaining useful energy out of nuclear reactions?
In 1928 the Russian-American physicist George Gamow (1904-1968) suggested that protons might be used as bombarding agents in place of alpha particles. Protons were only one-fourth as massive as alpha particles and the collision might be correspondingly less effective; on the other hand, protons had only half the positive charge of alpha particles and would not be as strongly repelled by the nuclei. Then, too, protons were much more easily available than alpha particles. To get a supply of protons one only had to ionize the very common hydrogen atoms, i.e., get rid of the single electron of the hydrogen atom, and a single proton is left.
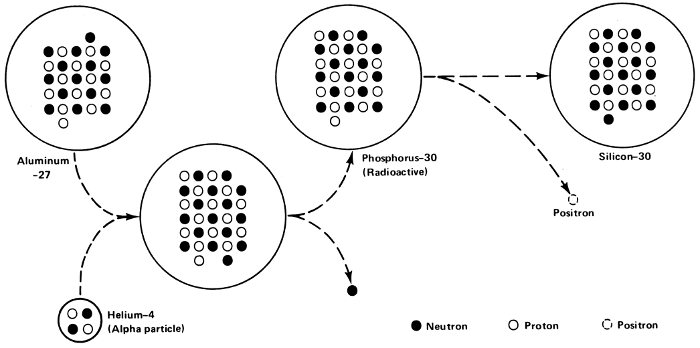
Artificial radioactivity.
Of course, protons obtained by the ionization of hydrogen atoms have very little energy, but could energy be imparted to them? Protons carry a positive charge and a force can therefore be exerted upon them by an electric or magnetic field. In a device that makes use of such fields, protons can be accelerated (made to go faster and faster), and thus gain more and more energy. In the end, if enough energy is gained, the proton could do more damage than the alpha particle, despite the former’s smaller mass. Combine that with the smaller repulsion involved and the greater ease of obtaining protons—and the weight of convenience and usefulness would swing far in the direction of the proton.
Physicists began to try to design “particle accelerators” and the first practical device of this sort was produced in 1929 by the two British physicists John Douglas Cockcroft (1897-1967) and Ernest Thomas Sinton Walton (1903- ). Their device, called an “electrostatic accelerator”, produced protons that were sufficiently energetic to initiate nuclear reactions. In 1931 they used their accelerated protons to disrupt the nucleus of lithium-7. It was the first nuclear reaction to be brought about by man-made bombarding particles.
Other types of particle accelerators were also being developed at this time. The most famous was the one built in 1930 by the American physicist Ernest Orlando Lawrence (1901-1958). In this device a magnet was used to make the protons move in gradually expanding circles, gaining energy with each lap until they finally moved out beyond the influence of the magnet and then hurtled out of the instrument in a straight line at maximum energy. This instrument was called a “cyclotron”.

Inventors of one of the first accelerators, Ernest T. S. Walton, left, and John D. Cockcroft, right, with Lord Ernest Rutherford at Cambridge University in the early 1930s.
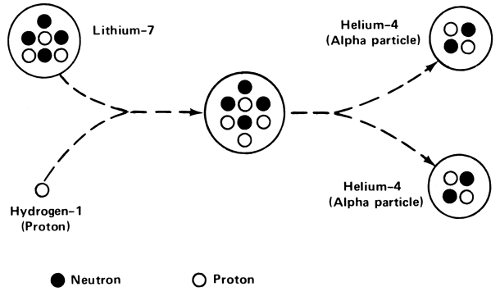
The bombardment of lithium-7 with protons was the first nuclear reaction caused by man-made particles.
The cyclotron was rapidly improved, using larger magnets and increasingly sophisticated design. There are now, at this time of writing, “proton synchrotrons” (descendants of that first cyclotron) that produce particles with over a million times the energy of those produced by Lawrence’s first cyclotron. Of course, the first cyclotron was only a quarter of a meter wide, while the largest today has a diameter of some 2000 meters.
As particle accelerators grew larger, more efficient, and more powerful, they became ever more useful in studying the structure of the nucleus and the nature of the subatomic particles themselves. They did not serve, however, to bring the dream of useful nuclear energy any closer. Though they brought about the liberation of vastly more nuclear energy than Rutherford’s initial bombardments could, they also consumed a great deal more energy in the process.
It is not surprising that Rutherford, the pioneer in nuclear bombardment, was pessimistic. To the end of his days (he died in 1937) he maintained that it would be forever impossible to tap the energy of the nucleus for use by man. Hopes that “nuclear power” might some day run the world’s industries were, in his view, an idle dream.

Ernest O. Lawrence holds a model of the first cyclotron in 1930, a year after its conception.
THE NEUTRON
Nuclear Spin
What Rutherford did not (and could not) take into account were the consequences of a completely new type of nuclear bombardment involving a type of particle unknown in the 1920s (though Rutherford speculated about the possibility of its existence).
The beginnings of the new path came about through the reluctant realization that there was a flaw in the apparently firmly grounded proton-electron picture of nuclear structure.
The flaw involved the “nuclear spin”. In 1924 the Austrian physicist Wolfgang Pauli (1900-1958) worked out a theory that treated protons and electrons as though they were spinning on their axes. This spin could be in either direction (or, as we would say in earthly terms, from west-to-east, or from east-to-west). Quantum theory has shown that a natural unit exists for what is called the angular momentum of this spin. Measured in terms of this natural unit of spin, the proton and the electron have spin ½. If the particle spun in one direction it was +½, if in the other it was -½.
When subatomic particles came together to form an atomic nucleus, each kept its original spin, and the nuclear spin was then equal to the total angular momentum of the individual particles that made it up.
For instance, suppose the helium nucleus is made up of 4 protons and 2 electrons, as was thought in the 1920s. Of the 4 protons, suppose that two had a spin of +½ and two of -½. Suppose also that of the 2 electrons, one had a spin of +½ and one of -½. All the spins would cancel each other. The total angular momentum would be zero.
Of course, it is also possible that all 6 particles were spinning in the same direction; all +½ or all -½. In that case the nuclear spin would be 3, either in one direction or the 93 other. If 5 particles were spinning in one direction and 1 in the other, then the total spin would be 2, in one direction or the other.
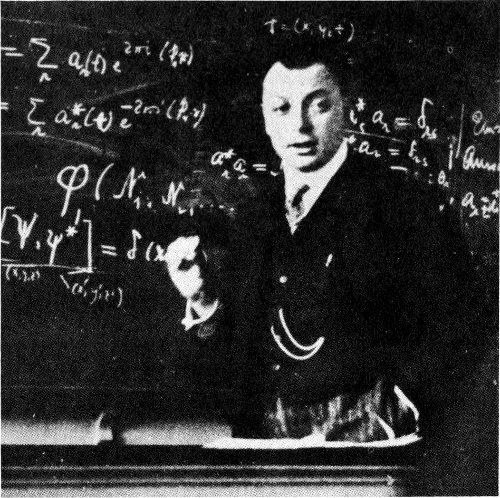
Wolfgang Pauli lecturing in Copenhagen in April 1929.
In short if you have an even number of particles in a nucleus, each with a spin of +½ or -½, then the total spin is either zero or a whole number, no matter what combination of positive and negative spins you choose. (The total spin is always written as a positive number.)
On the other hand, suppose you have lithium-7, which was thought to be made up of 7 protons and 4 electrons. If the 7 protons were all +½ and the 4 electrons were all -½ in their spins, the nuclear spin would be ⁷/₂ - ⁴/₂ = ³/₂.
If you have an odd number of particles in the nucleus, you will find that any combination of positive and negative spins will never give you either zero or a whole number as a sum. The sum will always include a fraction.
Consequently, if one measures the spin of a particular atomic nucleus one can tell at once whether that nucleus contains an even number of particles or an odd number.
This quickly raised a problem. The nuclear spin of the common isotope, nitrogen-14, was measured accurately over and over again and turned out to be 1. There seemed no doubt about that and it could therefore be concluded that there were an even number of particles in the nitrogen-14 nucleus.
And yet, by the proton-electron theory of nuclear structure, the nitrogen-14 nucleus, with a mass number of 14 and an atomic number of 7, had to be made up of 14 protons and 7 electrons for a total of 21 particles altogether—an odd number.
The nuclear spin of nitrogen-14 indicated “even number” and the proton-electron theory indicated “odd number”. One or the other had to be wrong, but which? The nuclear spin was a matter of actual measurement, which could be repeated over and over and on which all agreed. The proton-electron theory was only a theory. It was therefore the latter that was questioned.
What was to be done?
Suppose it is wrong to count protons and electrons inside the nucleus as separate particles. Was it possible that an electron and a proton, forced into the close confinement of the atomic nucleus might, by the force of mutual attraction, become so intimately connected as to count as a single particle. One of the first to suggest this, as far back as 1920, was Rutherford.
Such a proton-electron combination would be electrically neutral and in 1921 the American chemist William Draper Harkins (1873-1951) used the term “neutron” as a name for it.
If we look at the nitrogen-14 nucleus in this way then it is made up, not of 14 protons and 7 electrons, but of 7 protons and 7 proton-electron combinations. Instead of a total of 21 particles, there would be a total of 14; instead of an odd number, there would be an even number. The structure would now account for the nuclear spin.
But could such a revised theory of nuclear structure be made to seem plausible? The proton-electron theory seemed to make sense because both protons and electrons were known to exist separately and could be detected. If an intimate proton-electron combination could also exist, ought it not exist (or be made to exist) outside the nucleus and ought it not be detected as an isolated particle?
Discovery of the Neutron
Throughout the 1920s scientists searched for the neutron but without success.
One of the troubles was that the particle was electrically neutral. Subatomic particles could be detected in a variety of ways, but every single way (right down to the present time) makes use of their electric charge. The electric charge of a speeding subatomic particle either repels electrons or attracts them. In either case, electrons are knocked off atoms that are encountered by the speeding subatomic particle.
The atoms with electrons knocked off are now positively charged ions. Droplets of water vapor can form about these ions, or a bubble of gas can form, or a spark of light can be seen. The droplets, the bubbles, and the light can all be detected one way or another and the path of the subatomic particle could be followed by the trail of ions it left behind. Gamma rays, though they carry no charge, are a wave form capable of ionizing atoms.
All the particles and rays that can leave a detectable track of ions behind are called “ionizing radiation” and these are easy to detect.
The hypothetical proton-electron combination, however, which was neither a wave form nor a charged particle was not expected to be able to ionize atoms. It would wander among the atoms without either attracting or repelling electrons and would therefore leave the atomic structure intact. Its pathway could not be followed. In short, then, the neutron was, so to speak, invisible, and the search for it 96 seemed a lost cause. And until it was found, the proton-electron theory of nuclear structure, whatever its obvious deficiencies with respect to nuclear spin, remained the only one to work with.
Then came 1930. The German physicist Walther Wilhelm Georg Bothe (1891-1957) and a co-worker, H. Becker, were bombarding the light metal, beryllium, with alpha particles. Ordinarily, they might expect protons to be knocked out of it, but in this case no protons appeared. They detected some sort of radiation because something was creating certain effects while the alpha particles were bombarding the beryllium but not after the bombardment ceased.

Walther W. G. Bothe
To try to determine something about the properties of this radiation, Bothe and Becker tried putting objects in the way of the radiation. They found the radiation to be remarkably penetrating. It even passed through several centimeters of lead. The only form of radiation that was known at that time to come out of bombarded matter with the capacity of penetrating a thick layer of lead was gamma rays. Bothe and Becker, therefore, decided they had produced gamma rays and reported this.
In 1932 the Joliot-Curies repeated the Bothe-Becker work and got the same results. However, among the objects they placed in the path of the new radiation, they included paraffin, which is made up of the light atoms of carbon and hydrogen. To their surprise, protons were knocked out of the paraffin.
Gamma rays had never been observed to do this, but the Joliot-Curies could not think what else the radiation might be. They simply reported that they had discovered gamma rays to be capable of a new kind of action.

James Chadwick
Not so the English physicist James Chadwick (1891- ). In that same year he maintained that a gamma ray, which possessed no mass, simply lacked the momentum to hurl a proton out of its place in the atom. Even an electron was too light to do so. (It would be like trying to knock a baseball off the ground and into the air by hitting it with a ping-pong ball.)
Any radiation capable of knocking a proton out of an atom had to consist of particles that were themselves pretty massive. And if one argued like that, then it seemed that the radiation first observed by Bothe and Becker had to be the 98 long-sought-for proton-electron combination. Chadwick used Harkins’ term, neutron, for it and made it official. He gets the credit for the discovery of the neutron.
Chadwick managed to work out the mass of the neutron from his experiments and by 1934 it was quite clear that the neutron was more massive than the proton. The best modern data have the mass of the proton set at 1.007825, and that of the neutron just a trifle greater at 1.008665.
The fact that the neutron was just about as massive as the proton was to be expected if the neutron were a proton-electron combination. It was also not surprising that the isolated neutron eventually breaks up, giving up an electron and becoming a proton. Out of any large number of neutrons, half have turned into protons in about 12 minutes.
Nevertheless, although in some ways we can explain the neutron by speaking of it as though it were a proton-electron combination, it really is not. A neutron has a spin of ½ while a proton-electron combination would have a spin of either 0 or 1. The neutron, therefore, must be treated as a single uncharged particle.
The Proton-Neutron Theory
As soon as the neutron was discovered, the German physicist Werner Karl Heisenberg (1901- ) revived the notion that the nucleus must be made up of protons and neutrons, rather than protons and electrons. It was very easy to switch from the latter theory to the former, if one simply remembered to pair the electrons thought to be in the nucleus with protons and give the name neutrons to these combinations.
Thus, the helium-4 nucleus, rather than being made up of 4 protons and 2 electrons, was made up of 2 protons and 2 proton-electron combinations; or 2 protons and 2 neutrons. In the same way the oxygen-16 nucleus instead of being made up of 16 protons and 8 electrons, would be made up of 8 protons and 8 neutrons.
The proton-neutron theory would account for mass numbers and atomic numbers perfectly well. If a nucleus was made up of x protons and y neutrons, then the atomic number was equal to x and the mass number to x + y. (It is now possible to define the mass number of a nucleus in modern terms. It is the number of protons plus neutrons in the nucleus.)

Werner Heisenberg
The proton-neutron theory of nuclear structure could account for isotopes perfectly well, too. Consider the 3 oxygen isotopes, oxygen-16, oxygen-17, and oxygen-18. The 100 first would have a nucleus made up of 8 protons and 8 neutrons; the second, one of 8 protons and 9 neutrons; and the third, one of 8 protons and 10 neutrons. In each case the atomic number is 8. The mass numbers however would be 16, 17, and 18, respectively.
In the same way uranium-238 would have a nucleus built of 92 protons and 146 neutrons, while uranium-235 would have one of 92 protons and 143 neutrons.
By the new theory, can we suppose that it is neutrons rather than electrons that somehow hold the protons together against their mutual repulsion, and that more and more neutrons are required to do this as the nucleus grows more massive? At first the number of neutrons required is roughly equal to the number of protons. The helium-4 nucleus contains 2 protons and 2 neutrons, the carbon-12 nucleus contains 6 protons and 6 neutrons, the oxygen-16 nucleus contains 8 protons and 8 neutrons, and so on.
For more complicated nuclei, additional neutrons are needed. In vanadium-51, the nucleus contains 23 protons and 28 neutrons, five more than an equal amount. In bismuth-209, it is 83 protons and 126 neutrons, 43 more than an equal amount. For still more massive nuclei containing a larger number of protons, no amount of neutrons is sufficient to keep the assembly stable. The more massive nuclei are all radioactive.
The manner of radioactive breakdown fits the theory, too. Suppose a nucleus gives off an alpha particle. The alpha particle is a helium nucleus made up of 2 protons and 2 neutrons. If a nucleus loses an alpha particle, its mass number should decline by 4 and its atomic number by 2, and that is what happens.
Suppose a nucleus gives off a beta particle. For a moment, that might seem puzzling. If the nucleus contains only protons and neutrons and no electrons, where does the beta particle come from? Suppose we consider the neutrons 101 as proton-electron combinations. Within many nuclei, the neutrons are quite stable and do not break up as they do in isolation. In the case of certain nuclei, however, they do break up.
Thus the thorium-234 nucleus is made up of 90 protons and 144 neutrons. One of these neutrons might be viewed as breaking up to liberate an electron and leaving behind an unbound proton. If a beta particle leaves then, the number of neutrons decreases by one and the number of protons increases by one. The thorium-234 nucleus (90 protons, 144 neutrons) becomes a protactinium-234 nucleus (91 protons, 143 neutrons).
In short, the proton-neutron theory of nuclear structure could explain all the observed facts just as well as the proton-electron theory, and could explain the nuclear spins, which the proton-electron theory could not. What’s more, the isolated neutron had been discovered.
The proton-neutron theory was therefore accepted and remains accepted to this day.
The Nuclear Interaction
In one place, and only one, did the proton-neutron theory seem a little weaker than the proton-electron theory. The electrons in the nucleus were thought to act as a kind of glue holding together the protons.
But the electrons were gone. There were no negative charges at all inside the nucleus, only the positive charges of the proton, plus the uncharged neutron. As many as 83 positive charges were to be found (in the bismuth-209 nucleus) squeezed together and yet not breaking apart.
In the absence of electrons, what kept the protons clinging together?
Was it possible that the electrical repulsion between 2 protons is replaced by an attraction if those protons were pushed together closely enough? Can there be both an attraction and a repulsion, with the former the more 102 important at very short range? If this were so, that hypothetical attraction would have to have two properties. First, it would have to be extremely strong—strong enough to overcome the repulsion of two positive charges at very close quarters. Secondly, it would have to be short-range, for no attractive force between protons of any kind was ever detected outside the nucleus.
In addition, this short-range attraction would have to involve the neutron. The hydrogen-1 nucleus was made up of a single proton, but all nuclei containing more than 1 proton had to contain neutrons also to be stable, and only certain numbers of neutrons.
Until the discovery of the neutron, only two kinds of forces, or “interactions”, were known in the universe. These were the “gravitational interaction” and the “electromagnetic interaction”. The electromagnetic interaction was much the stronger of the two—trillions and trillions and trillions of times as strong as the gravitational attraction.
The electromagnetic attraction, however, includes both attraction (between opposite electric charges or between opposite magnetic poles) and repulsion (between like electric charges or magnetic poles). In ordinary bodies, the attractions and repulsions usually cancel each other entirely or nearly entirely, leaving very little of one or the other to be detected as surplus. The gravitational interaction, however, includes only attraction and this increases with mass. By the time you have gigantic masses such as the earth or the sun, the gravitational interaction between them and other bodies is also gigantic.
Both the gravitational and electromagnetic interactions are long-range. The intensity of each interaction declines with distance but only as the square of the distance. If the distance between earth and sun were doubled, the gravitational interaction would still be one-fourth what it is now. If the distance were increased ten times, the interaction would still be 1/(10 × 10) or 1/100 what it is now. It is for this 103 reason that gravitational and electromagnetic interactions can make themselves felt over millions of miles of space.
But now, with the acceptance of the proton-neutron theory of nuclear structure, physicists began to suspect the existence of a third interaction—a “nuclear interaction”—much stronger than the electromagnetic interaction, perhaps 130 times as strong. Furthermore, the nuclear interaction had to decline very rapidly with distance much more rapidly than the electromagnetic interaction did.
In that case, protons in virtual contact, as within the nucleus, would attract each other, but if the distance between them was increased sufficiently to place one outside the nucleus, the nuclear interaction would decrease in intensity to less than the electromagnetic repulsion. The proton would now be repelled by the positive charge of the nucleus and would go flying away. That is why atomic nuclei have to be so small; it is only when they are so tiny that the nuclear interaction can hold them together.
In 1932 Heisenberg tried to work out how these interactions might come into being. He suggested that attractions and repulsions were the result of particles being constantly and rapidly exchanged by the bodies experiencing the attractions and repulsions. Under some conditions, these “exchange particles” moving back and forth very rapidly between 2 bodies might force those bodies apart; under other conditions they might pull those bodies together.
In the case of the electromagnetic interaction, the exchange particles seemed to be “photons”, wave packets that made up gamma rays, X rays, or even ordinary light (all of which are examples of “electromagnetic radiation”). The gravitational interaction would be the result of exchange particles called “gravitons”. (In 1969, there were reports that gravitons had actually been detected.)
Both the photon and the graviton have zero mass and there is a connection between that and the fact that electromagnetic interaction and gravitational interaction decline 104 only slowly with distance. For a nuclear interaction, which declines very rapidly with distance, the exchange particle (if any) would have to have mass.
In 1935 the Japanese physicist Hideki Yukawa (1907- ) worked out in considerable detail the theory of such exchange particles in order to decide what kind of properties the one involved in the nuclear interaction would have. He decided it ought to have a mass about 250 times that of an electron, which would make it about ¹/₇ as massive as a proton. Since this mass is intermediate between that of an electron and proton, such particles eventually came to be called “mesons” from a Greek word meaning “intermediate”.
Once Yukawa published his theory, the search was on for the hypothetical mesons. Ideally, if they existed within the nucleus, shooting back and forth between protons and neutrons, there ought to be some way of knocking them out of the nucleus and studying them in isolation. Unfortunately, the bombarding particles at the disposal of physicists in the 1930s possessed far too little energy to knock mesons out of nuclei, assuming they were there in the first place.
There was one way out. In 1911 the Austrian physicist Victor Francis Hess (1883-1964) had discovered that earth was bombarded from every side by “cosmic rays”. These consisted of speeding atomic nuclei (“cosmic particles”) of enormous energies—in some cases, billions of times as intense as any energies available through particles produced by mankind. If a cosmic particle of sufficient energy struck an atomic nucleus in the atmosphere, it might knock mesons out of it.
In 1936 the American physicists Carl David Anderson (1905- ) and Seth Henry Neddermeyer (1907- ), studying the results of cosmic-particle bombardment of matter, detected the existence of particles of intermediate mass. This particle turned out to be lighter than Yukawa had predicted; it was only about 207 times as massive as an electron. Much worse, it lacked other properties that Yukawa had predicted. It did not interact with the nucleus in the manner expected.

Hideki Yukawa

Victor F. Hess

C. D. Anderson
In 1947, however, the English physicist Cecil Frank Powell (1903-1969) and his co-workers, also studying cosmic-particle bombardment, located another intermediate-sized body, which had the right mass and all the other appropriate properties to fit Yukawa’s theories.
Anderson’s particle was called a “mu-meson”, soon abbreviated to “muon”. Powell’s particle was called a “pi-meson”, soon abbreviated to “pion”. With the discovery of the pion, Yukawa’s theory was nailed down and any lingering doubt as to the validity of the proton-neutron theory vanished.

C. F. Powell
(Actually, it turns out that there are two forces. The one with the pion as exchange particle is the “strong nuclear interaction”. Another, involved in beta particle emission, for instance, is a “weak interaction”, much weaker than the electromagnetic but stronger than the gravitational.)
The working out of the details of the strong nuclear interaction explains further the vast energies to be found resulting from nuclear reactions. Ordinary chemical reactions, with the electron shifts that accompany them, involve the electromagnetic interaction only. Nuclear energy, with the 107 shifts of the particles inside the nucleus, involves the much stronger nuclear interaction.
Neutron Bombardment
As soon as neutrons were discovered, it seemed to physicists that they had another possible bombarding particle of extraordinary properties. Since the neutron lacked any electric charge, it could not be repelled by either electrons on the outside of the atoms or by the nuclei at the center. The neutron was completely indifferent to the electromagnetic attraction and it just moved along in a straight line. If it happened to be headed toward a nucleus it would strike it no matter how heavy a charge that nucleus might have and very often it would, as a result, induce a nuclear reaction where a proton would not have been able to.

J. Robert Oppenheimer
To be sure, it seemed just at first that there was a disadvantage to the neutron’s lack of charge. It could not be accelerated directly by any device since that always depended on electromagnetic interaction to which the neutron was impervious.
There was one way of getting around this and this was explained in 1935 by the American physicist J. Robert Oppenheimer (1904-1967) and by his student Melba Phillips.
Use is made here of the nucleus of the hydrogen-2 (deuterium) nucleus. That nucleus, often called a “deuteron”, is made up of 1 proton plus 1 neutron and has a mass number of 2 and an atomic number of 1. Since it has a unit positive charge, it can be accelerated just as an isolated proton can be.
Suppose, then, that a deuteron is accelerated to a high energy and is aimed right at a positively charged nucleus. That nucleus repels the deuteron, and it particularly repels the proton part. The nuclear interaction that holds together a single proton and a single neutron is comparatively weak as nuclear interactions go, and the repulsion of the nucleus that the deuteron is approaching may force the proton out of the deuteron altogether. The proton veers off, but the neutron, unaffected, keeps right on going and, with all the energy it had gained as part of the deuteron acceleration, smashes into the nucleus.
Within a few months of their discovery, energetic neutrons were being used to bring about nuclear reactions.
Actually, though, physicists didn’t have to worry about making neutrons energetic. This was a hangover from their work with positively charged particles such as protons and alpha particles. These charged particles had to be energetic to overcome the repulsion of the nucleus and to smash into it with enough force to break it up.
Neutrons, however, didn’t have to overcome any repulsion. No matter how little energy they had, if they were correctly aimed (and some always were, through sheer chance) they would approach and strike the nucleus.
In fact, the more slowly they travelled, the longer they would stay in the vicinity of a nucleus and the more likely they were to be captured by some nearby nucleus through the attraction of the nuclear interaction. The influence of the nucleus in capturing the neutron was greater the slower the neutron, so that it was almost as though the nucleus were 109 larger and easier to hit for a slow neutron than a fast one. Eventually, physicists began to speak of “nuclear cross sections” and to say that particular nuclei had a cross section of such and such a size for this bombarding particle or that.
The effectiveness of slow neutrons was discovered in 1934 by the Italian-American physicist Enrico Fermi (1901-1954).
Of course, there was the difficulty that neutrons couldn’t be slowed down once they were formed, and as formed they generally had too much energy (according to the new way of looking at things). At least they couldn’t be slowed down by electromagnetic methods—but there were other ways.
A neutron didn’t always enter a nucleus that it encountered. Sometimes, if it struck the nucleus a hard, glancing blow, it bounced off. If the nucleus struck by the neutron is many times as massive as the neutron, the neutron bounced off with all its speed practically intact. On the other hand, if the neutron hits a nucleus not very much more massive than itself, the nucleus rebounds and absorbs some of the energy, so that the neutron bounces away with less energy than it had. If the neutron rebounds from a number of comparatively light nuclei, it eventually loses virtually all its energy and finally moves about quite slowly, possessing no more energy than the atoms that surround it.
(You can encounter this situation in ordinary life in the case of billiard balls. A billiard ball, colliding with a cannon ball, will just bounce, moving just as rapidly afterward as before, though in a different direction. If a billiard ball strikes another billiard ball, it will set the target ball moving and bounce off itself with less speed.)
The energy of the molecules in the atmosphere depends on temperature. Neutrons that match that energy and have the ordinary quantity to be expected at room temperature are called “thermal” (from a Greek word meaning “heat”) neutrons. The comparatively light nuclei against which the 110 neutrons bounce and slow down are “moderators” because they moderate the neutron’s energy.
Fermi and his co-workers were the first to moderate neutrons, produce thermal neutrons, and use them, in 1935, to bombard nuclei. He quickly noted how large nuclear cross sections became when thermal neutrons were the bombarding particles.
It might seem that hope could now rise in connection with the practical use of energy derived from nuclear reactions. Neutrons could bring about nuclear reactions, even when they themselves possessed very little energy, so output might conceivably be more than input for each neutron that struck. Furthermore because of the large cross sections involved, thermal neutrons missed far less frequently than high-energy charged particles did.
But there was a catch. Before neutrons could be used, however low-energy and however sure to hit, they had to be produced; and in order to produce neutrons they had to be knocked out of nuclei by bombardment with high-energy protons or some other such method. The energy formed by the neutrons was at first never more than the tiniest fraction of the energies that went into forming the neutrons in the first place.
It was as though you could indeed light a candle with a single match, but you still had to look through 300,000 useless pieces of wood before you found a match. The candle would still be impractical.
Even with the existence of neutron bombardment, involving low energy and high cross section, Rutherford could, with justice, feel right down to the time of his death that nuclear energy would never be made available for practical use.
And yet, among the experiments that Fermi was trying in 1934 was that of sending his neutrons crashing into uranium 111 atoms. Rutherford had no way of telling (and neither had Fermi) that this, finally, was the route to the unimaginable.
FOOTNOTES
Quotation Credit
| Inside front cover | Copyright © by Abelard-Shuman, Ltd., New York. Reprinted by permission from Inside the Atom, Isaac Asimov, 1966. |
Photo Credits
| Cover | Thorne Films |
| Page facing inside front cover | The “Horsehead” Nebula in Orion, Hale Observatories. |
| Author’s Photo | Jay K. Klein |
| Contents pages | Lick Observatory |
| 68 | Dr. Erwin W. Mueller, The Pennsylvania State University |
| 70 | Yerkes Observatory |
| 86 | From Discovery of the Elements, Mary E. Weeks, Chemical Education Publishing Company, 1968. |
| 89 | The Central Press Photos, Ltd., and Sir John Cockcroft |
| 91 | Ernest Orlando Lawrence Livermore Laboratory |
| 93 | Samuel A. Goudsmit |
| 96 & 97 | Nobel Institute |
| 99 | Copyright © 1965 by Barbara Lovett Cline, reprinted from her volume The Questioners: Physicists and the Quantum Theory by permission of Thomas Y. Crowell, Inc., New York. |
| 105 & 106 | Nobel Institute |
| 107 | Alan W. Richards |
★ U.S. GOVERNMENT PRINTING OFFICE: 1975——640—285/14
A word about ERDA....
The mission of the U. S. Energy Research & Development Administration (ERDA) is to develop all energy sources, to make the Nation basically self-sufficient in energy, and to protect public health and welfare and the environment. ERDA programs are divided into six major categories:
· CONSERVATION OF ENERGY—More efficient use of existing energy sources, development of alternate fuels and engines for automobiles to reduce dependence on petroleum, and elimination of wasteful habits of energy consumption.
· FOSSIL ENERGY—Expansion of coal production and the development of technologies for converting coal to synthetic gas and liquid fuels, improvement of oil drilling methods and of techniques for converting shale deposits to usable oil.
· SOLAR, GEOTHERMAL, AND ADVANCED ENERGY SYSTEMS—Research on solar energy to heat, cool, and eventually electrify buildings, on conversion of underground heat sources to gas and electricity, and on fusion reactors for the generation of electricity.
· ENVIRONMENT AND SAFETY—Investigation of health, safety, and environmental effects of the development of energy technologies, and research on management of wastes from energy production.
· NUCLEAR ENERGY—Expanding medical, industrial and research applications and upgrading reactor technologies for the generation of electricity, particularly using the breeder concept.
· NATIONAL SECURITY—Production and administration of nuclear materials serving both civilian and military needs.
ERDA programs are carried out by contract and cooperation with industry, university communities, and other government agencies. For more information, write to USERDA—Technical Information Center, P. O. Box 62, Oak Ridge, Tennessee 37830.

United States
Energy Research and Development Administration
Office of Public Affairs
Washington, D.C. 20545

